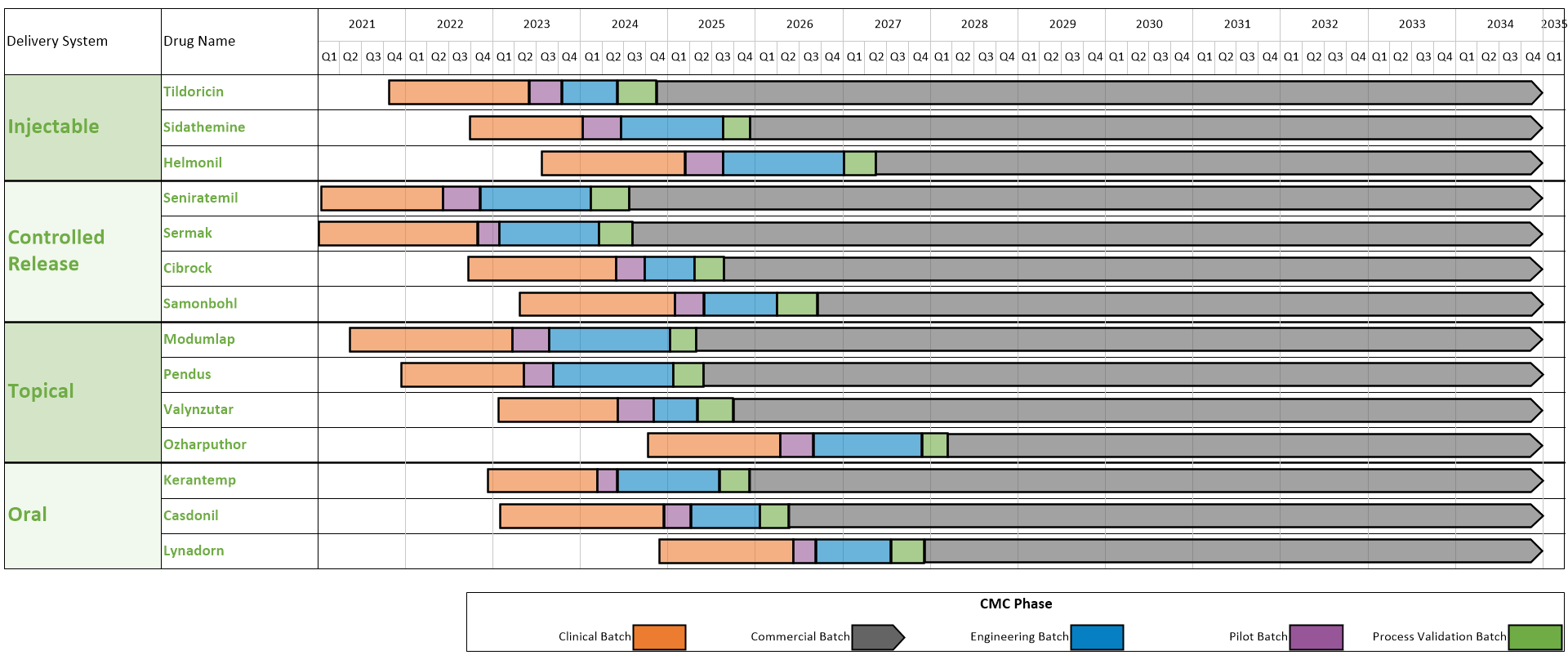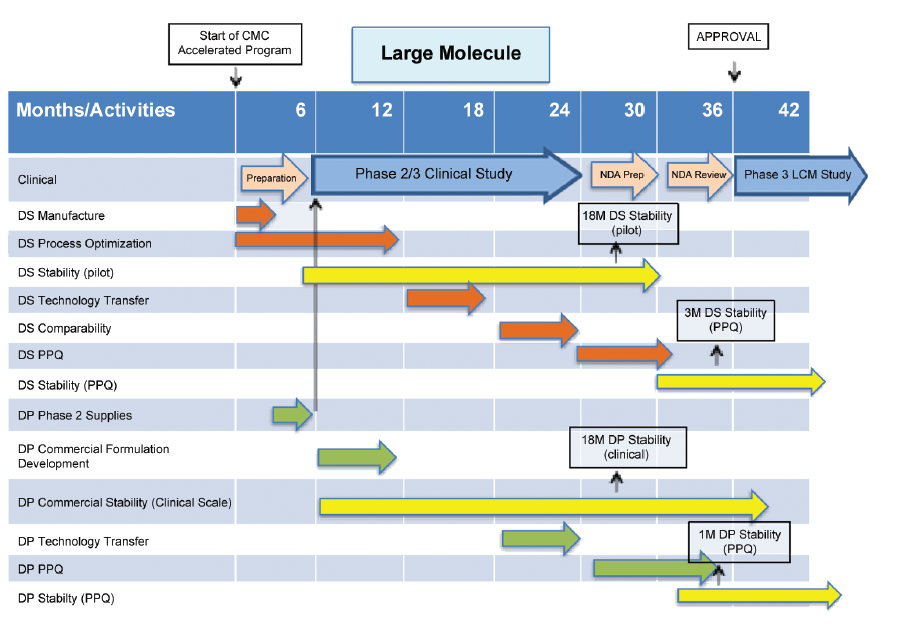
CMC Considerations when a Drug Development Project is Assigned Breakthrough Therapy Status | Pharmaceutical Engineering

CMC Development Strategies for Small Pharma | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

Technology Transfer of CMC Activities for MAb Manufacturing - BioProcess InternationalBioProcess International
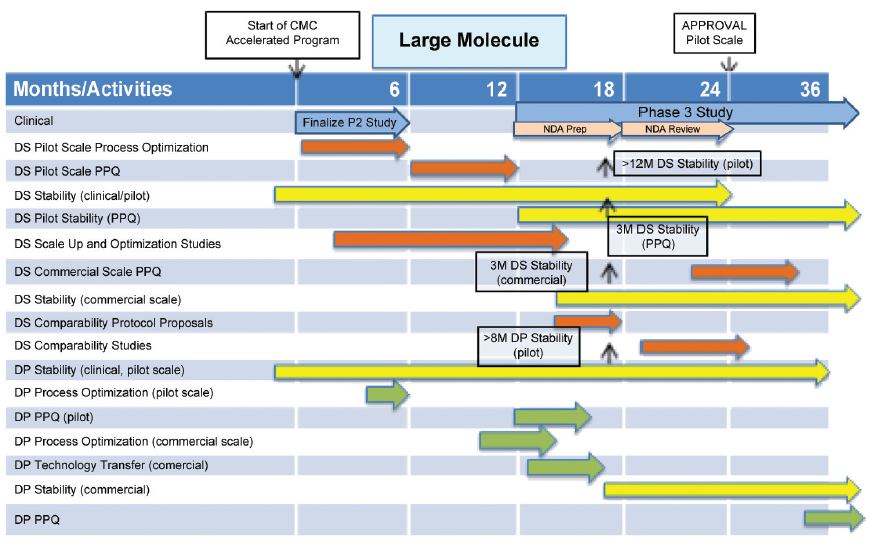
CMC Considerations when a Drug Development Project is Assigned Breakthrough Therapy Status | Pharmaceutical Engineering
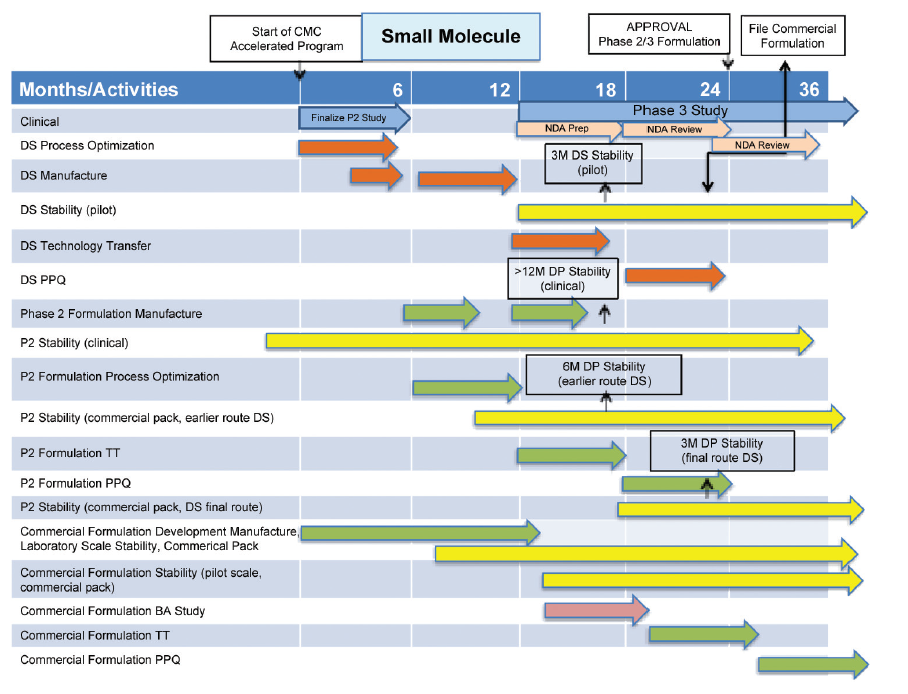
CMC Considerations when a Drug Development Project is Assigned Breakthrough Therapy Status | Pharmaceutical Engineering

SciRhom starts CMC development of its first drug candidate for clinical development | World Pharma Today
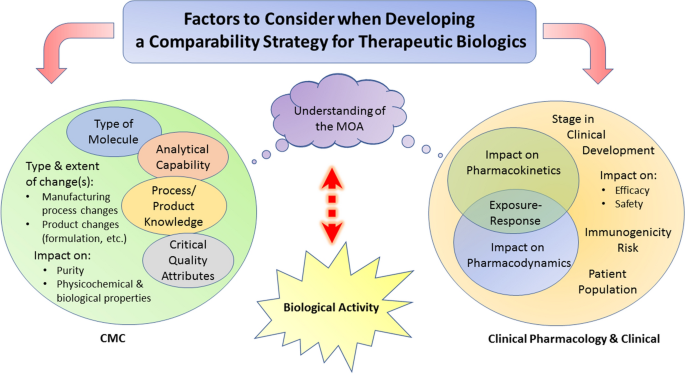
Comparability Considerations and Challenges for Expedited Development Programs for Biological Products | SpringerLink
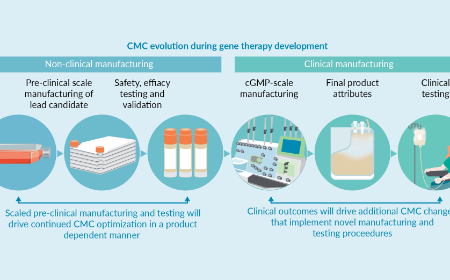
BioInsights - The Chemistry Manufacturing and Controls (CMC) section of gene therapy-based INDs: overview in a changing landscape

Phase-Appropriate CMC Activities Facilitate the Transition from Early Development through Commercialization

Phase-Appropriate Frameworks at the Intersection of CMC and cGMP Pathways | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

Regulatory tools to consider and integrate into overall US development... | Download Scientific Diagram
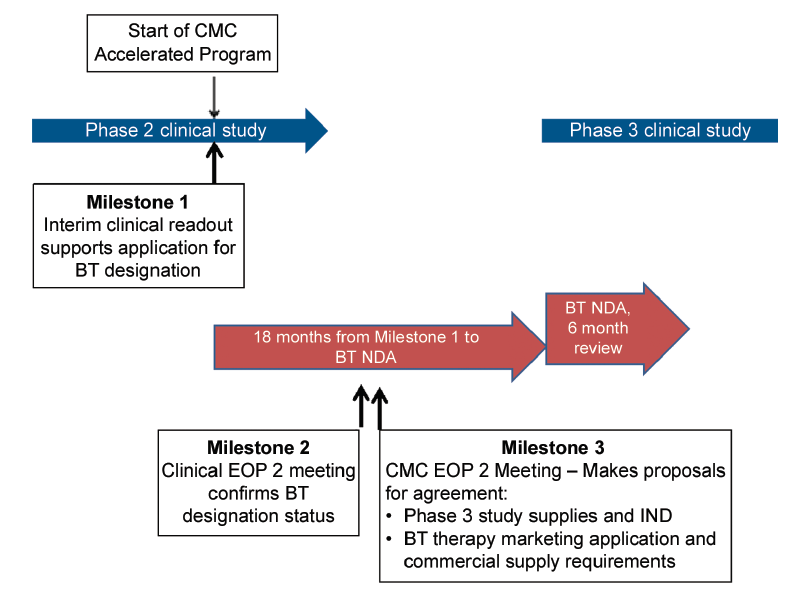
CMC Considerations when a Drug Development Project is Assigned Breakthrough Therapy Status | Pharmaceutical Engineering